S. African president receives first vaccine shot as immunization starts
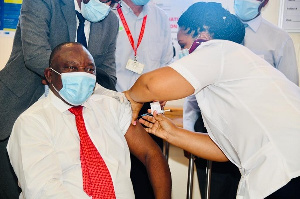
South Africa will administer its first COVID-19 vaccine on Wednesday, with President Cyril Ramaphosa to get the Johnson & Johnson (J&J) shot alongside health workers.
The first shot will be given at Khayelitsha District Hospital in Cape Town this afternoon, the government said.
Ramaphosa said in a statement that 80,000 J&J doses were being prepared for distribution across the country and that he would be inoculated to demonstrate his government’s confidence in the vaccine.
The J&J vaccine is being rolled out initially as a research study to further evaluate it in the field, with up to 500,000 health workers set to be immunised.
Ramaphosa said most vaccination centres would be ready to begin the country’s immunisation programme on Wednesday.
“The Johnson & Johnson vaccine has been shown in extensive trials to be safe and efficacious and will protect our health care workers from illness and death from COVID-19,” he said.
South Africa switched to the one-shot J&J vaccine after pausing the rollout of AstraZeneca’s two-dose vaccine this month.
Preliminary trial data showed the AstraZeneca shot developed in partnership with the University of Oxford offered minimal protection against mild to moderate illness from the country’s dominant coronavirus variant.
The decision to suspend the AstraZeneca rollout has added to scepticism among some South Africans about COVID-19 vaccines. The government says it could sell or swap its AstraZeneca doses.
South Africa has recorded about 1.5 million coronavirus infections and over 48,000 deaths, the most on the African continent. The government had initially planned to vaccinate 40 million people, or two-thirds of the population, to achieve some level of herd immunity, but it is not clear whether that target still stands.
Since late last year, South Africa has been battling a more infectious virus variant called 501Y.V2 that has alarmed health experts for its ability to potentially evade the immune response generated by prior exposure to the coronavirus or vaccines.
AstraZeneca says it believes its vaccine protects against severe COVID-19 and that it has started adapting it against the 501Y.V2 variant.
Source: reuters.com





